Swiss-based aQuaTox-Solutions offers animal-free alternative testing methods for chemicals in fish to replace animal experiments in research, product development and environmental risk assessment.
The idea came from the group of Professor Kristin Schirmer, co-founder of aQuaTox-Solutions and head of the Department of Environmental Toxicology at Eawag, the Swiss Federal Institute of Aquatic Science and Technology. The research of her team has been focusing on finding alternatives to animal testing for environmental risk assessment of chemicals and water samples with a focus on fish.
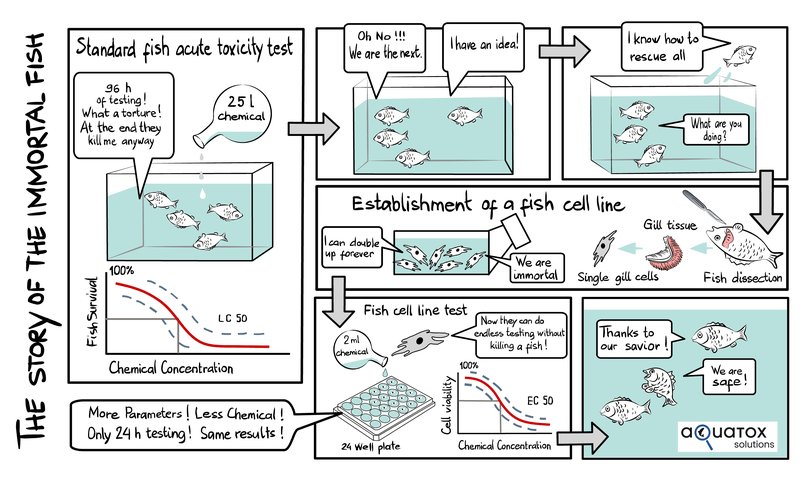
“The journey in the group of Kristin Schirmer started by developing a test that could replace the so-called acute fish toxicity test. In this test, about 42 fish are exposed to five concentrations of the test chemical; over four days, fish death is monitored. As you can imagine, by definition, this is a test that causes a lot of animal suffering. Yet, it is still the most frequently applied regulatory test with fish for assessing a chemical’s impact on the environment,” CEO Stephan Fischer explained. “The idea was to develop an assay procedure that is based on a fish gill cell line, which, in the way it is applied in this assay, essentially responds like a fish. As the word about this new method spread, we in Kristin’s team received requests from different parts of the world, asking if we could investigate certain chemicals and water samples using this new test procedure.”
However, since Eawag is a federally funded research institute and not a commercial service provider, the team could not provide the services and they decided to establish a company to offer gill cell line assays and other alternatives to fish tests as a service to clients worldwide.
The test
The method uses fish cells in culture to predict what would happen to these cells in fish – either by being damaged, for toxicological investigations, or by strengthening them, such as for studying new ingredients to boost fish health.

“For predicting acute fish toxicity with the fish gill cell line, for example, we let cells grow to form a dense epithelium and then expose this epithelium for one day to a range of concentrations of the test chemical or water sample in a simple exposure medium. We then apply a set of three fluorescent indicator dyes to determine the concentration that causes 50% of the cells to die, which can generally be taken at face value to predict the concentration that kills 50% of fish in the four-day acute fish toxicity test,” Fischer explained.
Another example is the use of a fish intestinal cell line to explore if feed additives strengthen or weaken the immune response, which is measured based on the regulation of a panel of genes. We can also use cells to predict the impact of chemical exposure on fish growth.
The fish cell lines were each isolated from healthy donor fish. When placed into a culture dish, with the right medium and given enough time, they developed into permanent cell lines. In other words, these cells can be cultured indefinitely and in this way provide a continuous source of fish cells for the tests.
“This not only saves the lives of thousands of fish but also a lot of resources. We need, for example, less than one-thousandth water and test chemicals compared to the conventional animal test; and we can obtain results much faster, often within a few days compared to weeks or even months when using fish. Overall, this, of course, also saves costs,” Fischer stated.
The team is combining in vitro tests with computer models to extrapolate the response of a cell population in a culture dish (in vitro) to a response in fish (in vivo) where needed. “Computer models essentially help us add information on fish physiology or the flow of information from one fish tissue to another, like filling a puzzle to form a full picture,” the team said.
Applications
The test based on rainbow trout gill cells was released by the Organisation for Economic Cooperation and Development (OECD) as a guideline in the field of environmental risk assessment. The “Fish Cell Line Acute Toxicity - The RTgill-W1 cell line assay” (OECD Testguideline 249) has become the first cell line-based, meaning animal-free, OECD test guideline for environmental risk assessment.
“The quality seal of OECD means that this assay is now globally accepted, no matter where it is carried out because it has been thoroughly standardized and proven to be robust and reproducible. This achievement is so important because it opens new avenues to truly replace fish in acute toxicity testing for pre-screening, product development, environmental hazard assessment and many other applications,” said Fischer.
The team also works with a cell line from rainbow trout liver for biotransformation and has plans to work with cells from the brain or testis. “Gill cells are the perfect model to quantify the toxicity of chemicals in the water. The intestinal cell line, on the other hand, is relevant for studying chemicals that enter the fish via feed or feed additives and ingredients. The brain cells could be of interest to measure neurotoxicity. And we are soon going to explore how far we can employ the rainbow trout testis cell line for reproductive effects,” Fischer said.
Testing feed additives
The team has tested a range of novel feed additives and ingredients to identify the most promising in terms of gut cell integrity, immune response and ultimately, fish intestinal health.
“In one case, we screened several additives to identify the most promising based on the response of the intestinal cells. When this additive was tested in a traditional fish feeding trial, it delivered the positive results that were expected based on the cell line test,” Fischer explained.
Another service was to test the toxicity of a novel, biodegradable fish bait to fish using the cell line approach.
Despite the fish feed industry being among the clients of the company, the methods are not yet well known in this area, the team said. “There lies tremendous potential in our approaches to save animals, time, money, etc. and even provide mechanistic insights that an animal experiment can hardly reveal, such as to what extent the intestinal epithelium acts as a barrier for transfer of chemicals or feed ingredients, how exactly the intestinal cells respond and how an additional bacterial or viral challenge can influence the intestinal barrier functionality. But also water quality assessment using the gill as the first site of contact in the fish could be an interesting application for the feed and aquaculture industry,” Fischer said.
Other services
Aside from its service portfolio, the company plans to add a product line to enable clients to perform certain analyses in-house, with support from aQuaTox-Solutions for data analysis and interpretation. “It is indeed our expert knowledge and our flexibility to accommodate a wide range of needs of our clients that are unique core assets of our enterprise,” Fischer concluded.



![Grassini_005[29]_0](/media/images/Grassini_00529_0.2e16d0ba.fill-300x300.jpg)








